IMPORTANT SAFETY INFORMATION
INDICATION & IMPORTANT SAFETY INFORMATION
Intracranial Hemorrhage—Serious intracranial hemorrhage (ICH) may occur with AYVAKIT treatment; fatal events occurred in <1% of patients. Overall, ICH (e.g., subdural hematoma, ICH, and cerebral hemorrhage) occurred in 2.9% of 749 patients who received AYVAKIT in clinical trials. In GIST patients, ICH occurred in 3 of 267 patients (1.1%) and two (0.7%) of the events were Grade ≥3 and resulted in discontinuation.
Monitor patients closely for risk factors of ICH which may include history of vascular aneurysm, ICH or cerebrovascular accident within the prior year, concomitant use of anticoagulant drugs, or thrombocytopenia. Symptoms of ICH may include headache, nausea, vomiting, vision changes, or altered mental status. Advise patients to seek immediate medical attention for signs or symptoms of ICH. Permanently discontinue AYVAKIT if ICH of any grade occurs.
Cognitive Effects—Cognitive adverse reactions can occur in patients receiving AYVAKIT and occurred in 33% of 995 patients overall in patients who received AYVAKIT in clinical trials including: 41% of 601 GIST patients (5% were Grade ≥3). Depending on the severity, withhold AYVAKIT and then resume at same dose or at a reduced dose upon improvement, or permanently discontinue.
Photosensitivity—AYVAKIT may cause photosensitivity reactions. In all patients treated with AYVAKIT in clinical trials (n=1049), photosensitivity reactions occurred in 2.5% of patients. Advise patients to limit direct ultraviolet exposure during treatment with AYVAKIT and for one week after discontinuation of treatment.
Embryo-Fetal Toxicity—AYVAKIT can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females and males of reproductive potential to use an effective contraception during treatment with AYVAKIT and for 6 weeks after the final dose of AYVAKIT. Advise women not to breastfeed during treatment with AYVAKIT and for 2 weeks following the final dose.
Adverse Reactions—The most common adverse reactions (≥20%) were edema, nausea, fatigue/asthenia, cognitive impairment, vomiting, decreased appetite, diarrhea, increased lacrimation, abdominal pain, constipation, rash, dizziness, and hair color changes.
Drug Interactions—Avoid coadministration of AYVAKIT with strong or moderate CYP3A inhibitors. If coadministration with a moderate CYP3A inhibitor cannot be avoided, reduce dose of AYVAKIT. Avoid coadministration of AYVAKIT with strong or moderate CYP3A inducers. If contraception requires estrogen, limit ethinyl estradiol to ≤20 mcg unless a higher dose is necessary.
To report suspected adverse reactions, contact Blueprint Medicines Corporation at 1-888-258-7768 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
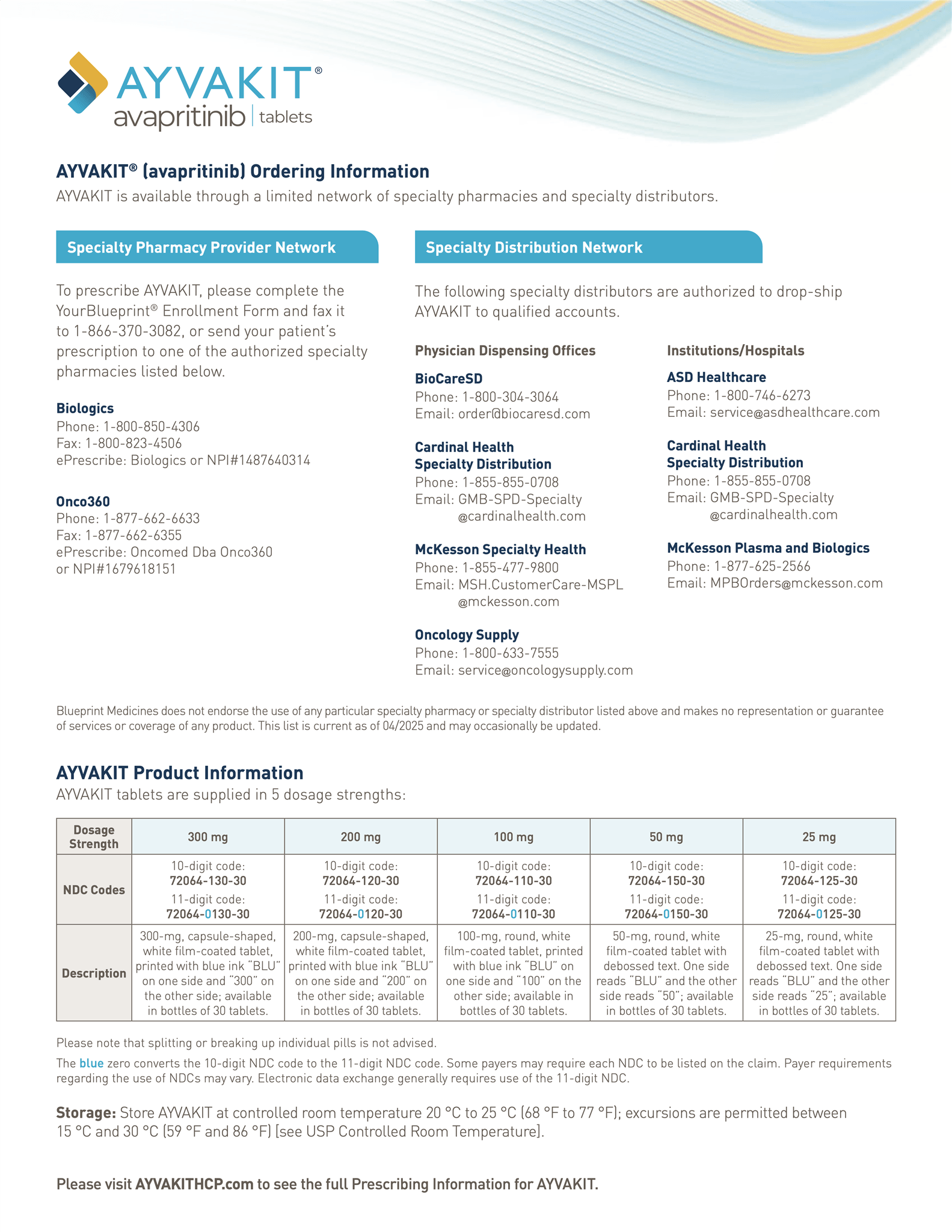
AYVAKIT is available in 100-mg, 200-mg, and 300-mg tablets.
Please click here to see the full Prescribing Information for AYVAKIT.
References:
- AYVAKIT [prescribing information]. Cambridge, MA: Blueprint Medicines Corporation; November 2024.
- Evans EK et al. Sci Transl Med. 2017;9(414):eaao1690.
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Gastrointestinal Stromal Tumors V.2.2024. © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Accessed September 18, 2024. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
- Casali PG et al. Ann Oncol. 2018;29(4):iv68-iv78.